Rising acidity in the world’s oceans could have dire consequences for millions, experts warn
- Carbon dioxide seeping into the ocean from volcanoes create pockets of acidity
- These mimic future conditions in the ocean if fossil fuel burning continues
- The studies show increased acidity majorly affects wild fish and marine life
- Creating a chain of effects weakening ocean reefs and disruptions to fishing
Rising acidity in the world’s oceans due to fossil fuel burning could have disastrous consequences for millions who depend on the seas for their livelihoods, new research has warned.
Scientists studying pockets of ocean acidity created by carbon dioxide seeping from volcanoes found that it negatively impacted marine life in Japan and wild fish populations in the Mediterranean.
These acidic conditions are what experts expect in the future if carbon dioxide levels continue to rise in the atmosphere due to the burning of fossil fuels.
Impacts on marine life can affect the wider food chain and fishing industries – as well as global coastal populations that feed off the ocean or relies on its tourism.

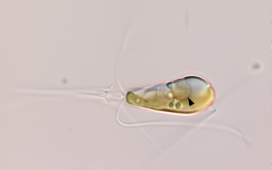
Ocean acidification could have disastrous consequences for millions globally who depends on it for their livelihood, new research has warned. The photo shows Professor Jason Hall-Spencer conducting a survey off the coast of Japan
The studies were conducted by the University of Plymouth and Tsukuba’s Shimoda Marine Research Centre in Japan.
They were based around the coast of Japan, where rising ocean acidity – known as acidification – is having a major impact on marine life, and in the Mediterranean where it was affecting the wild fish population.
Both regions have volcanic carbon dioxide seeps, where carbon dioxide bubbles out of active volcanic openings underwater and dissolves into the sea making it acidic.
These seeps have shown that reefs made by organisms with shells or skeletons- including oysters and corals – are sensitive to ocean acidification.
They also degraded reefs provide less coastal protection and less habitat for commercially important fish and shellfish.
This increases the risks to goods and services that are ocean already affected by climate change.
It could lead to shifts in seaweed dominance, habitat degradation and a loss of biodiversity including in many coastal regions.

Pockets of ocean acidity created by carbon dioxide seeping from volcanoes have allowed scientists to study the consequences of acidification on marine life and the coastal populations
Dr Ben Harvey, Assistant Professor at the University of Tsukuba’s Shimoda Marine Research Centre, said: ‘We are releasing around 1 million tons of carbon dioxide per hour into the Earth’s atmosphere.
‘About 25 per cent of this gas is taken up by the ocean where it reacts with seawater to form a weak acid, causing surface ocean pH to fall by around 0.002 units per year.
‘The chemistry of this rapid change in surface waters is understood, yet there is uncertainty about its effects on society which is what we are trying to overcome in this study.’
Professor Jason Hall-Spencer, Professor of Marine Biology at the University of Plymouth and the report’s lead author, added: ‘The Paris Agreement on climate change was welcome.
‘But it does not mention ocean acidification, nor the fact that this rapid change in surface ocean chemistry undermines the social, economic and environmental pillars of sustainable development.
‘The time is ripe for a ‘Paris Agreement for the oceans’, with the specific target to minimise and address the impacts of ocean acidification, including through enhanced scientific cooperation at all levels.’
The full report is published in Emerging topics in Life Sciences.